Australia’s National Greenhouse Accounts are made up of a series of comprehensive reports and databases that estimate and account for Australia’s greenhouse gas emissions. The National Inventory Report fulfils Australia’s inventory reporting requirements under the United Nations Framework Convention on Climate Change (UNFCCC) and its Kyoto Protocol.
The National Inventory Report contains national greenhouse gas emission estimates from 1990 to the current year of reporting.
The Global Warming Potential for each GHG is used and is regularly updated. The current values are (2020) are:
- 1 tonne of carbon dioxide (CO2) = 1 tonne of CO2 equivilent (e-)
- 1 tonne of Methane (CH4) = 28 tonnes ofCO2 e-
- 1 tonne of Nitrous Oxide (N20) = 265 tonnes of CO2 e-
Western Australia's Agricultural Emissions (2019)
Sectors reported in National Inventory (2019) and WA proportions
- Energy (92%)
- Industrial Processes (5%)
- Agriculture (11%)
- Land Use, Land-Use Change and Forestry (-9%)
- Waste (2%)
Western Australia's emissions from agriculture over time
WA's GHG emisions have decreased steadily since 1990, mainly due to a decrease in livestock numbers. Livestock contribute the largest portion of emissions through enteric fermentation, that is methane produced by the rumen and released via respiration (belching). Agricultural soils are the next biggest contributor via the release of nitrous oxide (N02).
Source: the National GHG Inventory for emissions, DPIRD, ABS for production. Analysis by DPIRD.
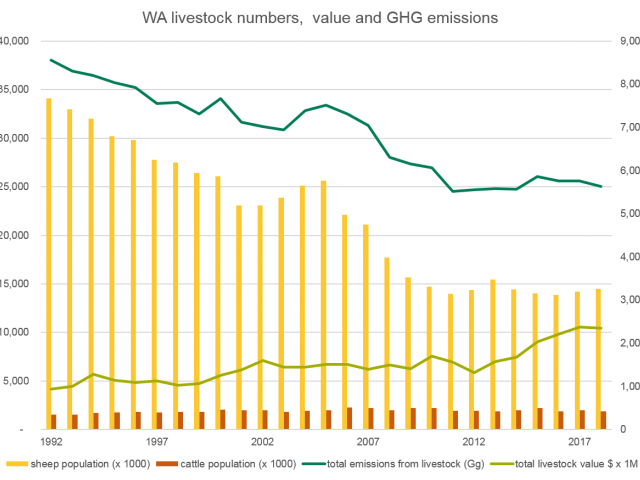
Western Australia's Livestock Emissions (2019)
Source: the National GHG Inventory for emissions, DPIRD, ABS for production. Analysis by DPIRD.
The GHG emissions are shown against the relative size of the WA beef herd and sheep flock. The lime green line shows the relative value of the industry over the same time period.
Western Australia's Cropping Emissions (2019)
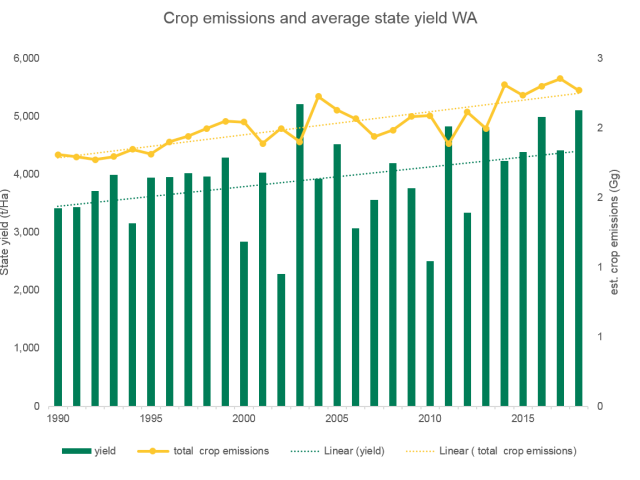
Source: the National GHG Inventory for emissions, DPIRD, ABS for production. Analysis by DPIRD.
The GHG emissions for sectors that influence the emissions for cropping in WA include agricultural soils under horticulture. The emissions are shown alongside the total yield (tonnes per hectare of grain) for the state over the same period.
Definitions
Enteric fermentation
Enteric fermentation is a natural part of the digestive process in ruminant animals such as cattle, sheep, goats, and buffalo. Microbes in the rumen decompose and ferment food, producing methane as a by-product. Calculated by the number of animals x class x feed intake x factor for methane.
Manure management
Manure acts as an emission source for both methane and nitrous oxide, and the quantity emitted is linked to environmental conditions, type of management and composition of the manure. Organic matter and nitrogen content are the main things influencing emission of methane and nitrous oxide, respectively.
Calculated by the number of animals x feed intake x factor for methane. NO2 for pasture grazing is added in agricultural soils.
Agricultural soils
Direct and indirect emissions of nitrous oxide from soils arise from microbial and chemical transformations that produce and consume nitrous oxide in the soil. The transformations involve inorganic nitrogen mainly ammonium, nitrite and nitrate. Nitrogen compounds can be added to the soil through:
- the application of inorganic nitrogen fertilisers
- the application of animal wastes to pastures
- the application of crop residues
- mineralisation due to loss of soil carbon and cultivation of organic soils
- atmospheric nitrogen deposition
- leaching of N from soils and surface runoff, and subsequent denitrification in rivers and estuaries (Areas with evapotranspiration ratios less than 0.8 or more than 1 are deemed areas where leaching and runoff occurs).
Field burning of residues
The burning of residual crop material releases CH4, N2O, CO, NOx and NMVOCs into the atmosphere. CO2 is not included in the calculations as is assumed that an equivalent amount of CO2 was removed by the growing crop.
Liming
Adding carbonates to soils in the form of lime (eg. calcic limestone (CaCO3 ) or dolomite (CaMg(CO3 )2 )) results in CO2 emissions, as the carbonate reacts with acids in the soil to produce bicarbonate and eventually leading to the production of CO2 and water.
Urea application
Adding urea to soils for fertilisation leads to a loss of the CO2 that was fixed during the manufacturing process. Similar to the reaction following the addition of lime, the bicarbonate that is formed evolves into CO2 and water.